
About us
Tackling the toughest cancers.
Head on.
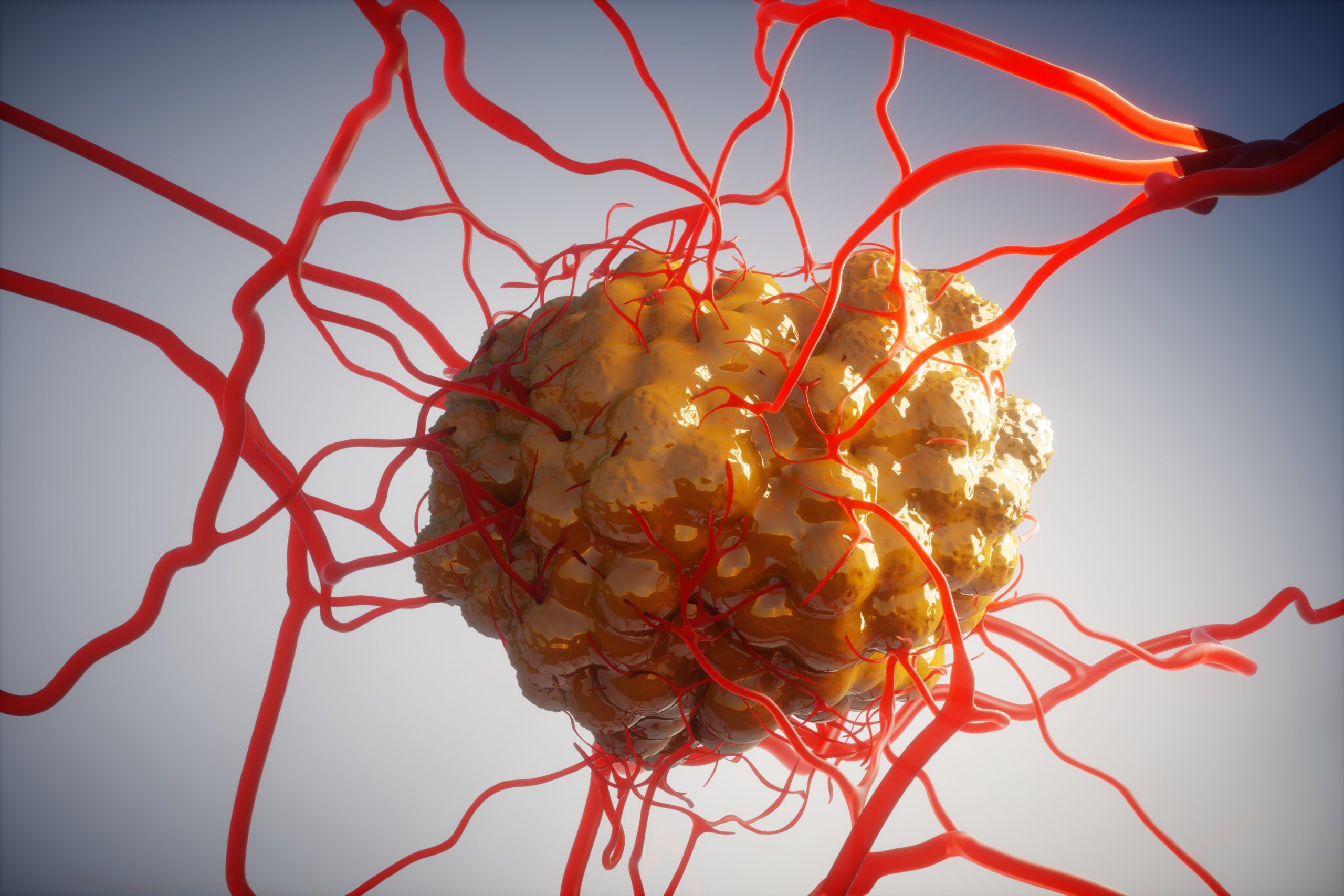
Penetration of anti-cancer agents into and throughout solid tumours is widely recognised as presenting a major limitation to their effectiveness.
SonoTran® is being developed to provide a step-change in the therapeutic index of anti-cancer agents without the development costs or delays associated with drug reformulation. SonoTran® is designed to increase the dose and distribution of anti-cancer agents within solid tumours, thereby increasing the efficacy and/or reducing the toxicity of these agents across the majority of the most challenging solid tumour cancers.
Our team.


Dr Matthew Frohn
Board Member
Prof. Constantin Coussios OBE FREng
Board Member and Co-Founder
George Robinson
Board Member
David Preston
Finance Director
Dr Cliff Rowe
Development Director
Dr Calum Crake
Technical Director
Prof. Fergus Gleeson FRCP FRCR
Clinical Advisor
Prof. Hardev Pandha MB ChB (Birm) FRACP FRCP PhD CSST (Medical Oncology)
Clinical Advisor
Mr Robert Jones
Clinical AdvisorOur values.

Our story.
OxSonics was founded in 2013 to commercialise ground-breaking technological advances developed over more than ten years at the Biomedical Ultrasonics, Biotherapy and Biopharmaceuticals Laboratory (BUBBL) at the University of Oxford in the UK. BUBBL is an internationally-recognized centre of excellence in therapeutic ultrasound and drug delivery within the Oxford Institute of Biomedical Engineering.
Since 2013, OxSonics has built a world-leading multi-disciplinary product development and commercialisation team that have successfully developed the SonoTran® Platform as a scalable commercial-grade product. We work closely with both medical oncologists and oncology radiologists to ensure our products are directed at the specific patient groups that stand to benefit most but also that our products fit seamlessly into existing patient care pathways in an oncology ward setting.
Today, SonoTran® is being evaluated clinically and we’re working with multiple partners to tackle the toughest cancers, head on.
Our supporters. Our collaborators.
Longwall Ventures


Oxford Technology and Innovations EIS Fund

MMK Capital

Perivoli Innovations

University of Oxford

Wellcome

Innovate UK

Oxford University Hospitals NHS Foundation Trust

